Abstract
Tyrosine kinase inhibitors (TKIs) are commonly believed to provide almost-normal lifespans to patients with chronic-phase (CP) chronic myeloid leukemia (CML). This is, however, not supported by Surveillance, Epidemiology, and End Results (SEER) mortalities >10 years after diagnoses that reveal a CP-CML relative risk (RR) of death of 2.14 (95% confidence interval [CI], 1.97-2.32); normal lifespans yield RR = 1.00, and by 10 years after diagnoses, most CML patients still alive were in CP at diagnosis, dates of which were between 2001 and 2009. This CP-CML RR estimate of 2.14 is comparable to those of Hodgkin's lymphoma (2.08; 95% CI, 1.95-2.22) and acute myeloid leukemia (2.22; 95% CI, 1.99-2.47) and higher than those of non-Hodgkin's lymphoma (1.62; 95% CI, 1.59-1.65) and chronic lymphocytic leukemia (1.84; 95% CI, 1.79-1.9). Thus, CP-CML is not functionally cured. In contrast, the RRs of hairy cell leukemia (1.14; 95% CI, 0.98-1.31) and acute promyelocytic leukemia (1.15; 95% CI, 0.87-1.49) are consistent with these leukemias being functionally cured. Of excess CP-CML deaths, roughly two thirds were by disease. These excess deaths by disease may be due in part to prolonged dose interruptions in response to treatment-related toxicities or to reduce financial burdens (ie, absence of TKI therapy). Frequencies of such events, and thus deaths by disease, could be decreased by reducing TKI doses moderately as cancer loads fall low enough to pose negligible progression risks. We present CML-intrinsic, TKI-agnostic support for reducing TKI doses when BCR::ABL1IS is <1% and decreasing.
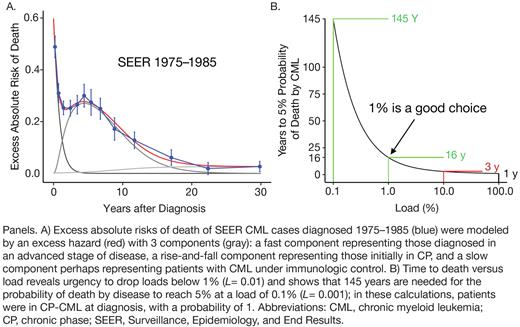
Times from diagnosis to death for 2732 SEER patients with CML diagnosed between 1975 and 1985 (before the availability of TKIs) informed a Markov model of CP → accelerated phase → blast crisis → death that assumed CML-saturated marrows in the absence of disease-modifying therapies during this time. The probabilities of transitioning from each phase to the next were fitted to the excess absolute risk of death (compared with the general US population) as a function of years after diagnosis (Panel A). To predict TKI effects on the probability of death by disease, the chronic-to-accelerated phase transition rate was assumed to be proportional to TKI-induced reductions in CML load fraction L, due to the reduced number of leukemic cells susceptible to a transforming mutation. Reducing L by a factor of 10 (from 1 to 0.1) increased the amount of time needed to reach a 5% probability of death by disease from 1 to 3 years. Lowering L by an additional factor of 10 (to 0.01 [1%]) increased the expected number of years to 16 (ie, ~5-fold). Finally, a third reduction of L by a factor of 10 (to 0.001 [0.1%]) yielded nearly an additional 10-fold increase in the time needed to reach a 5% probability of death by disease, from 16 to 145 years (Panel B). Gains in relative delay in 5% probability of death by disease divided by relative decrease in L were maximal at L = 0.015, which can be argued to correspond to BCR::ABLIS = 0.75% = ~1%, which appears to be a point of departure in Panel B.
Based on data that predates TKIs, our analysis suggests that there is a strong need to lower disease burdens (BCR::ABLIS) below 1% and essentially no need to lower them below 0.1%. Thus, while it is important to use drug dosing that maintains responses at 1% or lower, if BCR::ABLIS is <1% and decreasing, dose reductions should be considered. Such reductions may prevent deaths associated with mismanagement of drug directly and deaths by disease indirectly, as reducing doses prospectively may avoid toxicities that may otherwise result in prolonged interruptions that cause to rise enough to cause death by disease. These dose reduction benefits should be included in patient-physician discussions of pursuing lower BCR::ABLIS levels in hopes of achieving a treatment-free remission versus pursuing lower doses to mitigate toxicity risks. Envisioned here is a future in which personalized patient safety parameters are weighed against personalized predictions of BCR::ABLIS response dynamics that, through computer simulations incorporating our Markov model, project to risks of progression and death by disease. Decision support based on such models may help patients and physicians balance drug versus disease risks.
Disclosures
Radivoyevitch:Takeda: Research Funding. Druker:Astra-Zeneca: Other: Clinical Trial Funding, Research Funding; Recludix Pharma: Other: Sponsored Research Agreement ; Enliven Therapeutics: Other: Sponsored Research Agreement ; VB Therapeutics: Other: Founder; Multicancer Early Detection Consortium: Membership on an entity's Board of Directors or advisory committees; Beat AML LLS: Other: Joint Steering Committee ; CureOne: Membership on an entity's Board of Directors or advisory committees; Burroughs Wellcome Fund: Membership on an entity's Board of Directors or advisory committees; Vincerx Pharma: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Recludix Pharma: Current holder of stock options in a privately-held company, Other: Scientific Advisory Board ; Amgen: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; GRAIL: Current holder of stock options in a privately-held company, Other: Scientific Advisory Board ; Iterion Therapeutics: Current holder of stock options in a privately-held company, Other: Scientific Advisory Board ; Enliven Therapeutics: Current holder of stock options in a privately-held company, Other: Scientific Advisory Board ; Blueprint Medicines: Current holder of stock options in a privately-held company, Other: Scientific Advisory Board ; Aptose Biosciences: Current holder of stock options in a privately-held company, Other: Scientific Advisory Board ; RUNX1 Research Program: Other: Scientific Advisory Board ; Novartis: Other: Scientific Advisory Board; Clinical Trial Funding , Patents & Royalties: 6958335 (exclusive license), Research Funding; Nemucore Medical Innovations: Other: Scientific Advisory Board ; DNA SEQ: Other: Scientific Advisory Board ; Celgene: Other: Scientific Advisory Board ; Cepheid: Other: Scientific Advisory Board ; Therapy Architects (ALLCRON): Other: Scientific Advisory Board ; Adela Bio: Other: Scientific Advisory Board ; Aileron Therapeutics: Other: Scientific Advisory Board ; US Patent: Patents & Royalties: 4326534; US Patent: Patents & Royalties: 7416873; US Patent: Patents & Royalties: 6958335; US Patent: Patents & Royalties: 7592142; US Patent: Patents & Royalties: 10473667; US Patent: Patents & Royalties: 10664967; US Patent: Patents & Royalties: 11049247; Merck: Patents & Royalties: Monoclonal antiphosphotyrosine antibody 4G10; CytoImage: Patents & Royalties: QD Molecular Assay for Personalized Oncoprotein Detection in Leukemia (exclusive license); Sun Pharma Advanced Research Company: Patents & Royalties: Mutated ABL Kinase Domains (non-exclusive license); (Novartis exclusive license) and OHSU and Dana-Farber Cancer Institute (one Merck exclusive license, one CytoImage, Inc. exclusive license, and one Sun Pharma Advanced Research Company non-exclusive license): Patents & Royalties: Patent 6958335. Radich:HIG: Consultancy; NuProbe, Cepheid, Novartis, Amgen: Membership on an entity's Board of Directors or advisory committees. Kantarjian:AbbVie: Honoraria, Research Funding; ImmunoGen: Research Funding; Ascentage: Membership on an entity's Board of Directors or advisory committees, Research Funding; Astellas Health: Honoraria, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Consultancy, Research Funding; Ipsen Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Research Funding; KAHR Medical Ltd: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Research Funding; NOVA Research: Honoraria; Jazz Pharmaceuticals: Research Funding; Pfizer: Honoraria, Research Funding; Takeda: Honoraria. Tang:Takeda: Current Employment. Hanley:Takeda: Current Employment. Song:Takeda: Current Employment. Smith:Takeda: Current Employment. Bottino:Takeda Development Center Americas Inc.: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal